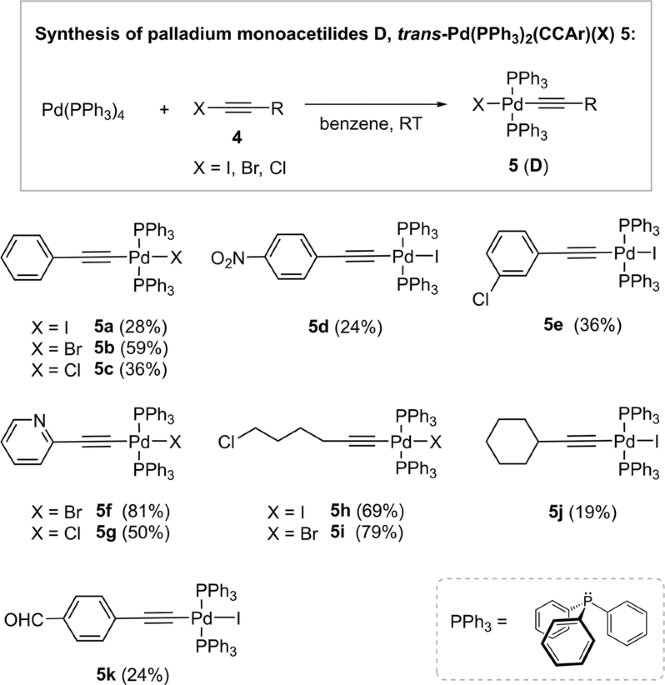
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry

Palladium-Catalyzed Aerobic Homocoupling of Alkynes: Full Mechanistic Characterization of a More Complex Oxidase-Type Behavior | ACS Catalysis

Palladium- and Nickel-Catalyzed Cross-Couplings of Unsaturated Halides Bearing Relatively Acidic Protons with Organozinc Reagents | The Journal of Organic Chemistry
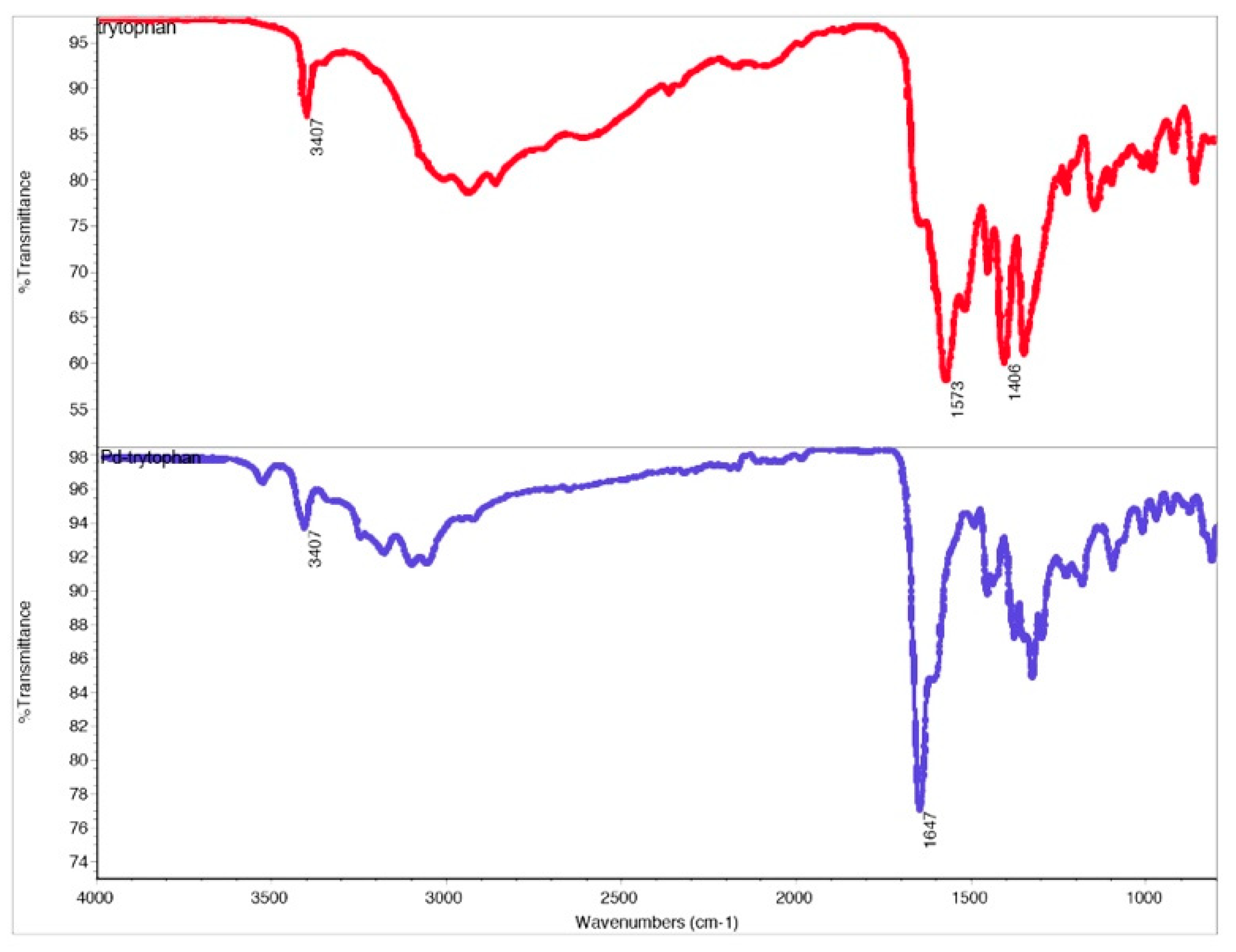
Molecules | Free Full-Text | Synthesis, Characterization, and Non-Covalent Interactions of Palladium(II)-Amino Acid Complexes

Anionic Palladium(0) and Palladium(II) Ate Complexes - Kolter - 2017 - Angewandte Chemie International Edition - Wiley Online Library
Novel reactions of metal-metal bonds. Insertion of isocyanides and carbon monoxide into the palladium-palladium bond of some palladium(I) dimers | Journal of the American Chemical Society
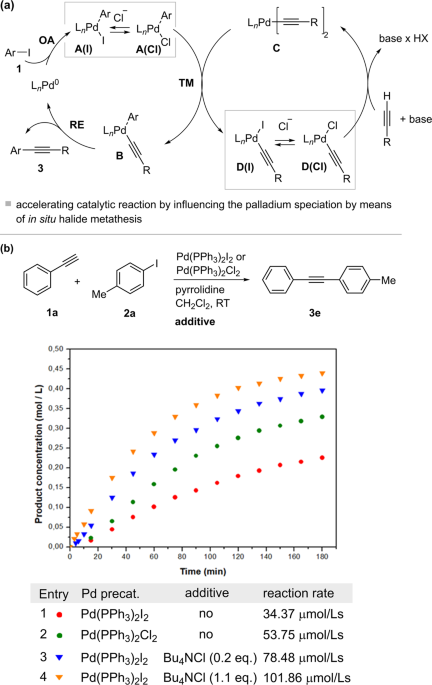
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry
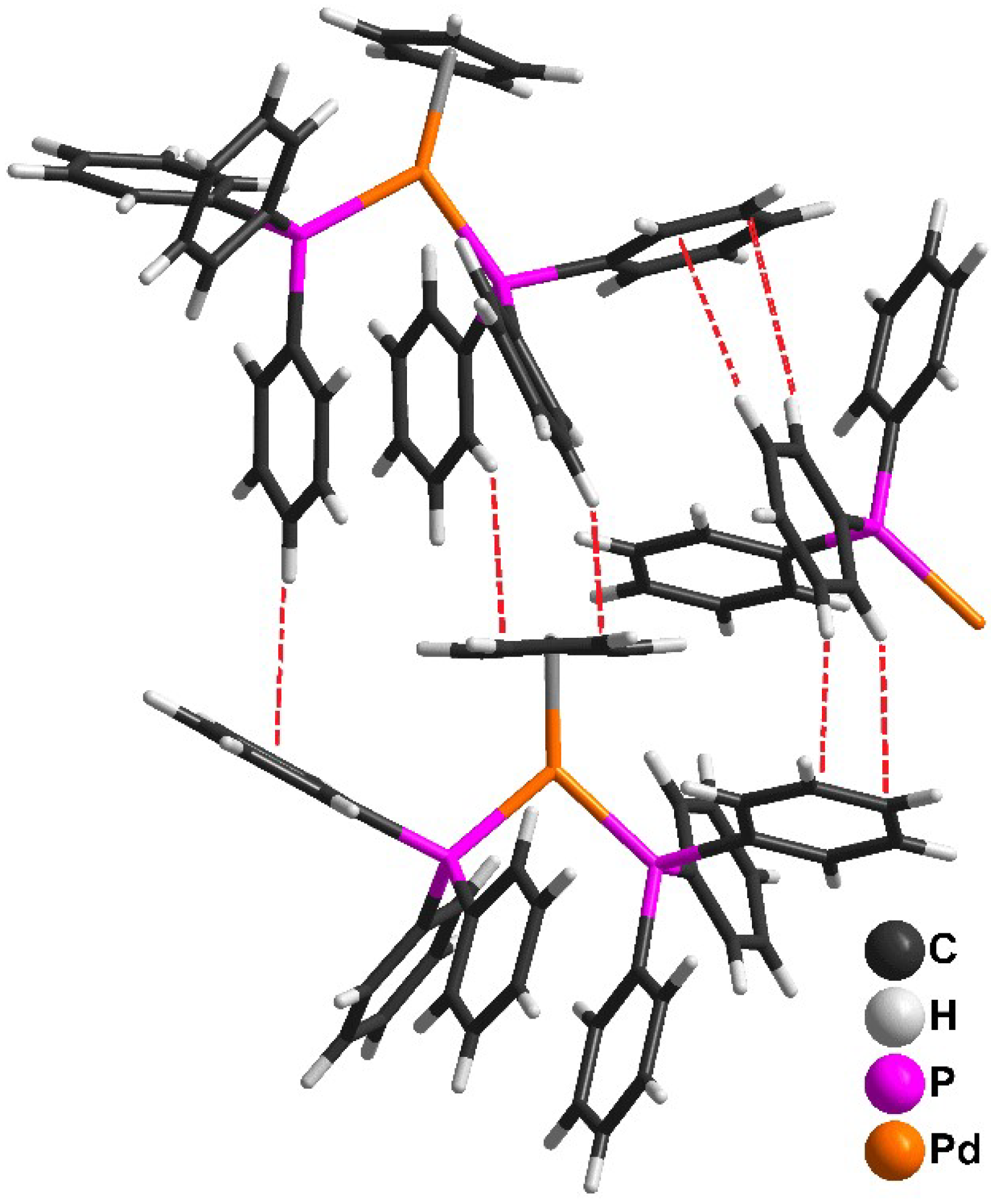
Molecules | Free Full-Text | Novel Route to Cationic Palladium(II)–Cyclopentadienyl Complexes Containing Phosphine Ligands and Their Catalytic Activities

Palladium‐Catalyzed Electrophilic Substitution via Monoallylpalladium Intermediates - Solin - 2003 - Angewandte Chemie - Wiley Online Library

Palladium N-Heterocyclic Carbene Complexes: Synthesis from Benzimidazolium Salts and Catalytic Activity in Carbon-carbon Bond-forming Reactions | Protocol (Translated to German)
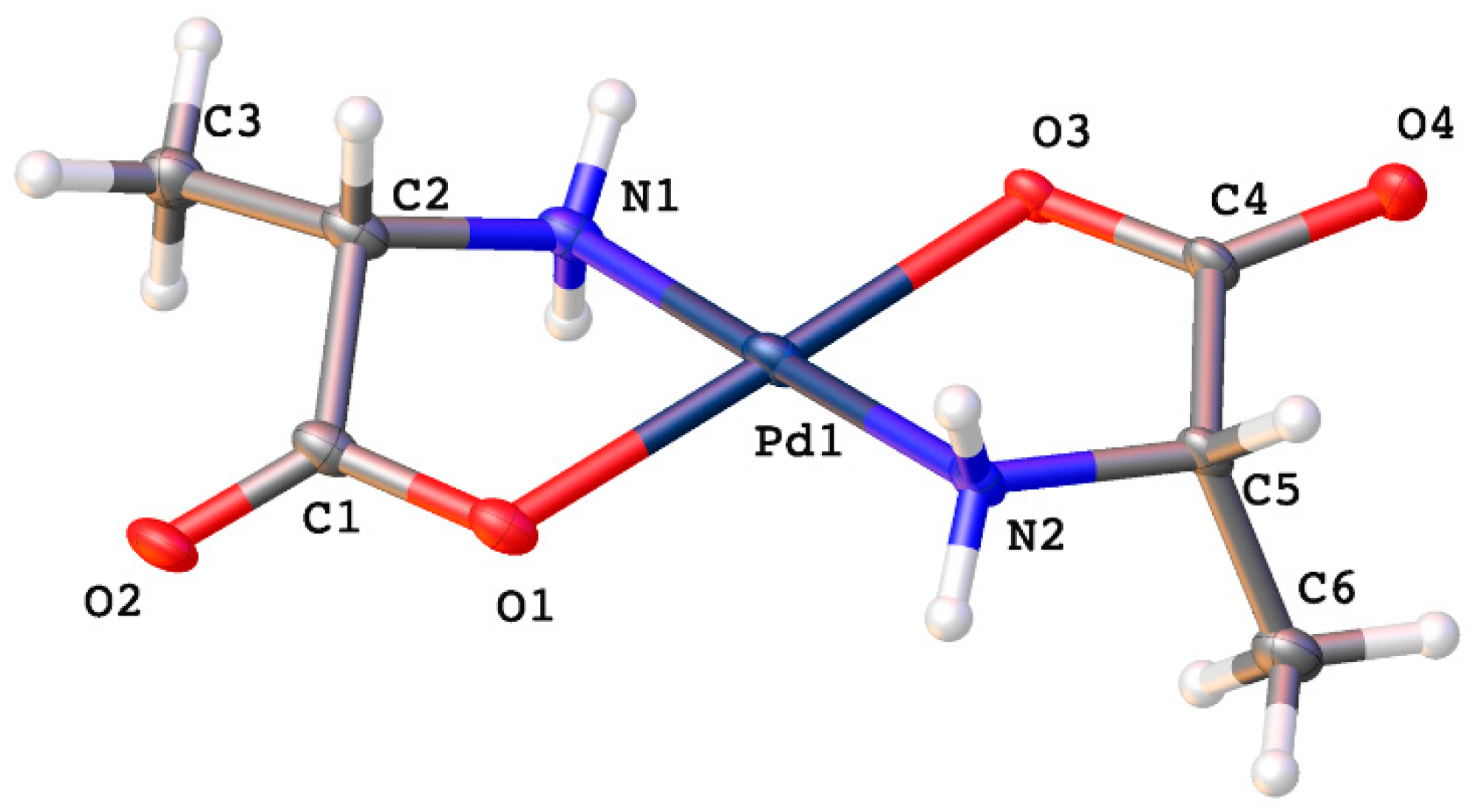
Molecules | Free Full-Text | Synthesis, Characterization, and Non-Covalent Interactions of Palladium(II)-Amino Acid Complexes
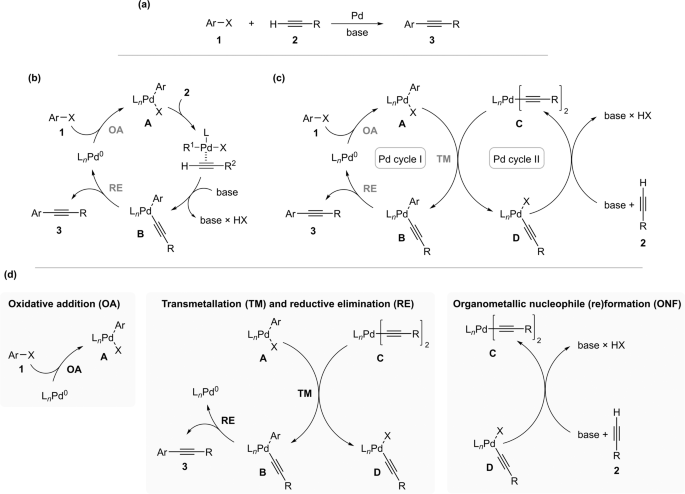
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry
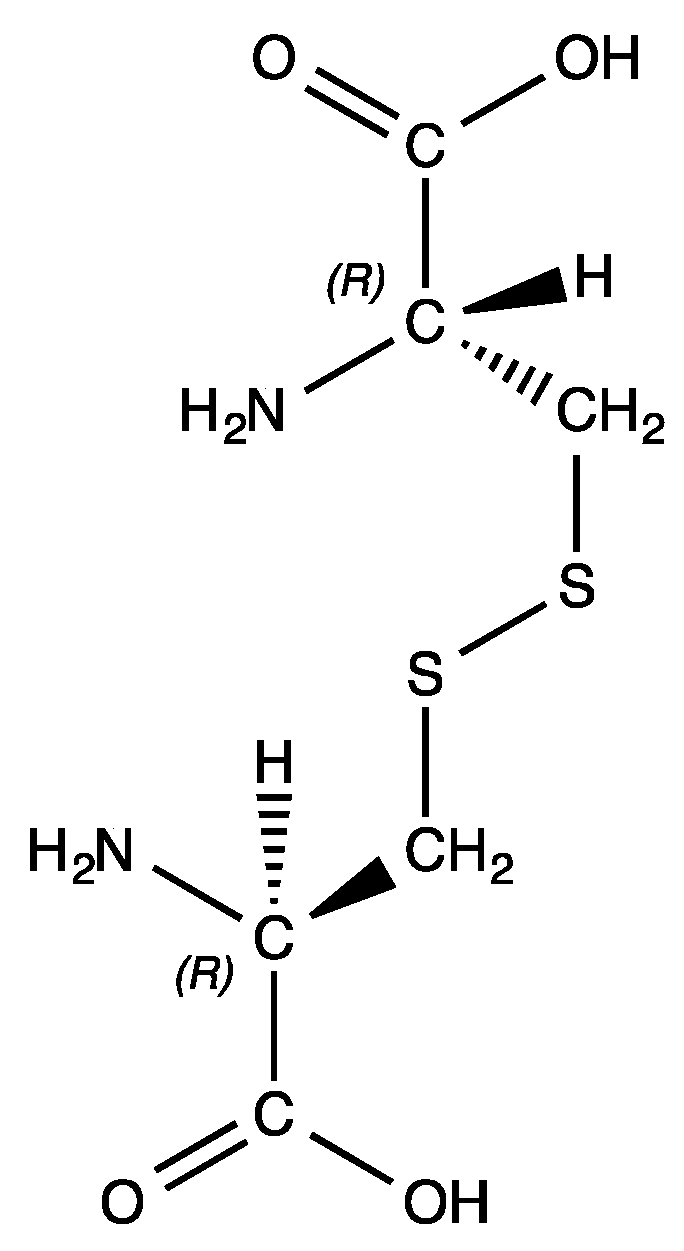
Molecules | Free Full-Text | Synthesis, Characterization, and Non-Covalent Interactions of Palladium(II)-Amino Acid Complexes

Carbene palladium precatalyst (A) and 2-oxo-3-methylbenzothiazole (B). | Download Scientific Diagram

Pd(0)-Catalyzed Phosphorus−Carbon Bond Formation. Mechanistic and Synthetic Studies on the Role of the Palladium Sources and Anionic Additives | Organometallics